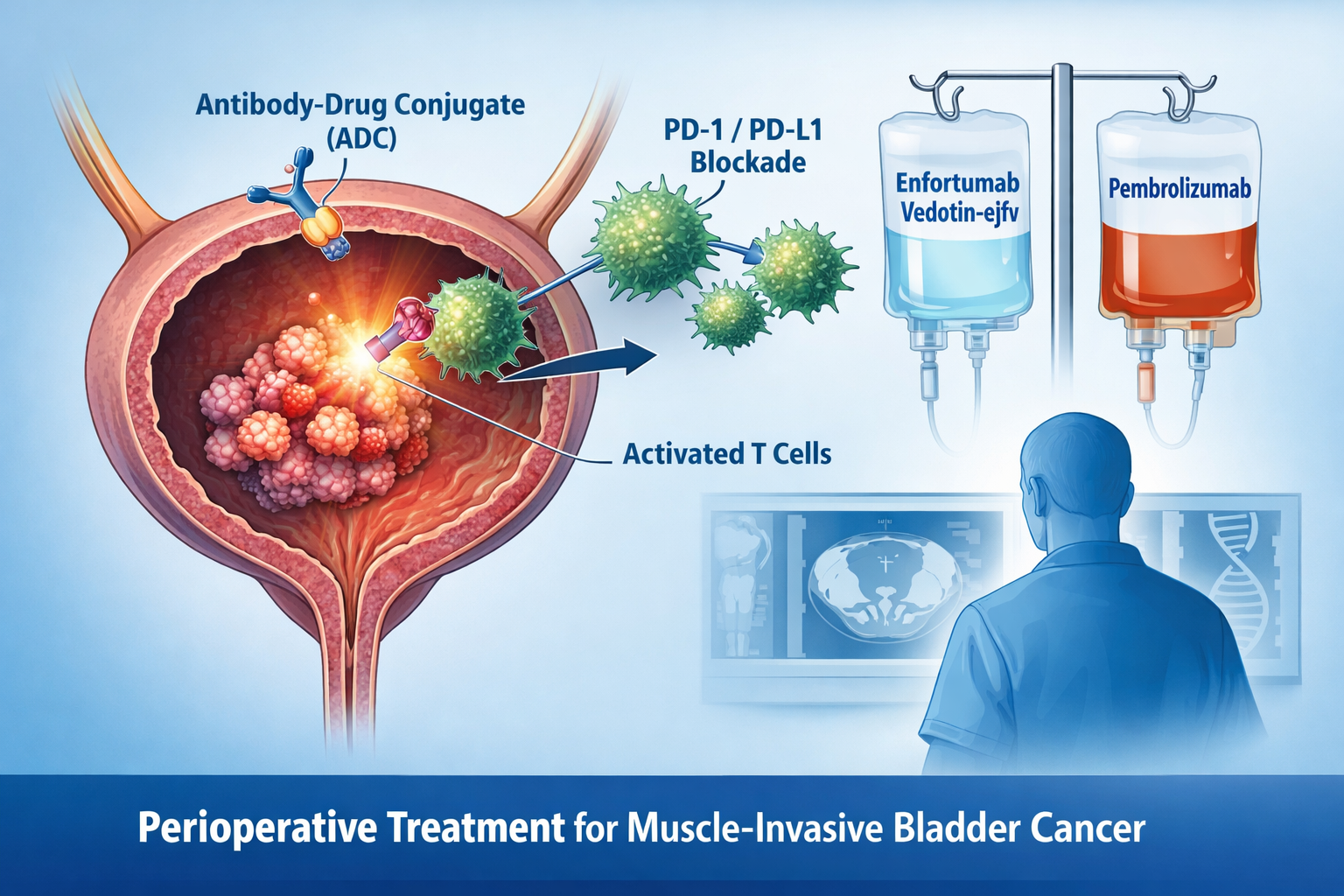
A New Era in MIBC: Perioperative Pembrolizumab + Enfortumab Vedotin Approved for Cisplatin-Ineligible Patients
On November 21, 2025, the U.S. Food and Drug Administration approved pembrolizumab (Keytruda or Keytruda Qlex) combined with enfortumab vedotin-ejfv (Padcev) as a perioperative therapy (neoadjuvant + adjuvant) for adults with muscle-invasive bladder cancer (MIBC) who are ineligible for cisplatin. This is a first-of-its-kind labeled treatment option in this setting.