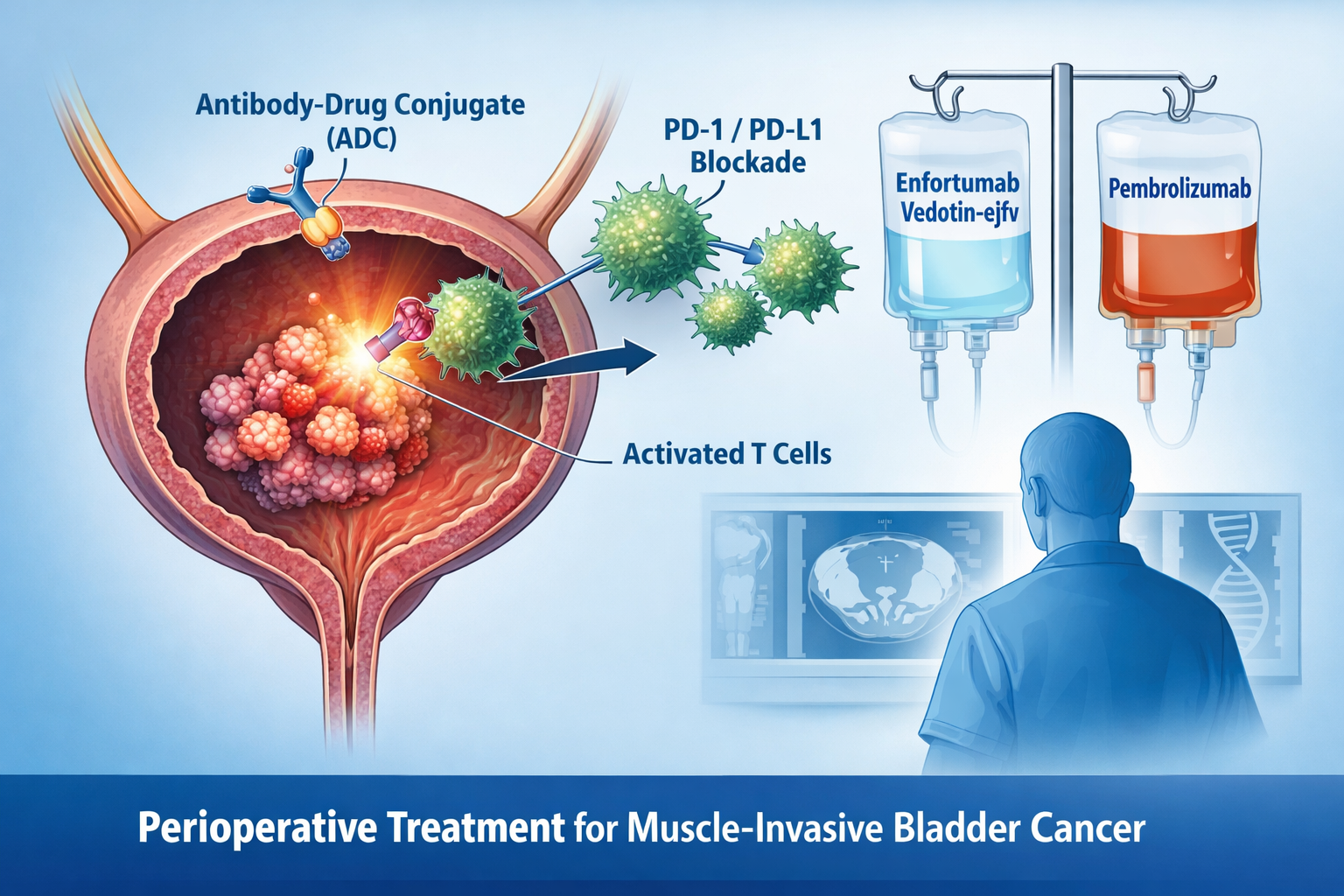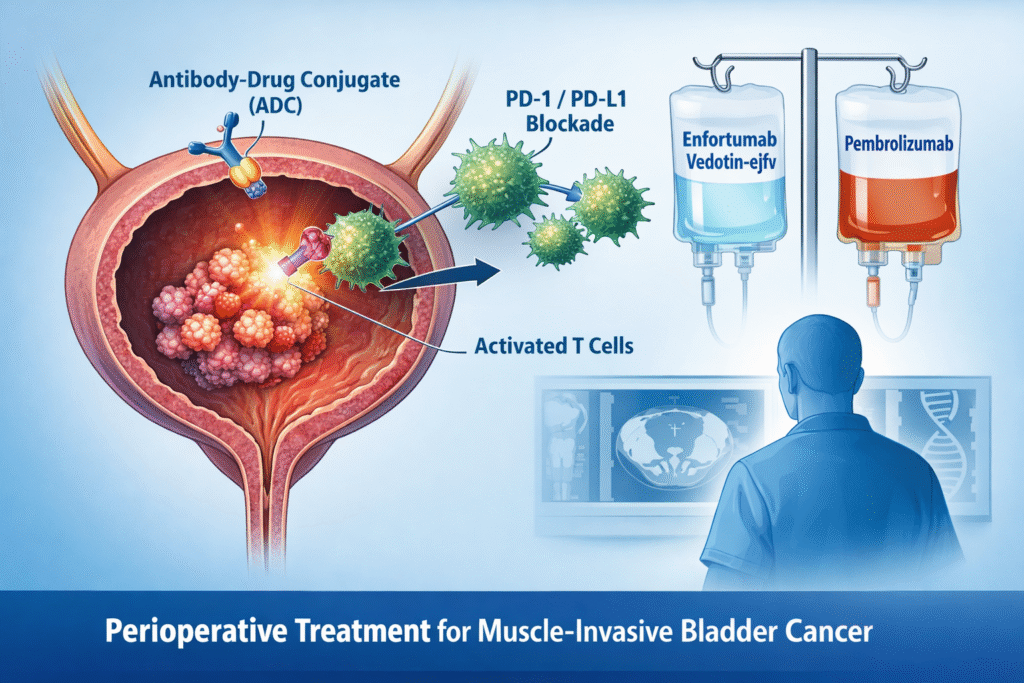
Phase III KEYNOTE-B15 data demonstrate significant improvements in event-free survival (EFS), overall survival (OS) and pathologic complete response (pCR) rates compared with standard cisplatin-based perioperative chemotherapy. The safety profile was manageable and consistent with the known effects of each agent.
Clinical Takeaway
Enfortumab vedotin-ejfv + pembrolizumab is a promising option for cisplatin-eligible patients with muscle-invasive bladder cancer (MIBC), demonstrating improvements in EFS, OS and pCR with a manageable safety profile.
Drug Profile & Mechanism
- Agent: enfortumab vedotin‑ejfv + pembrolizumab
- Class:
- enfortumab vedotin‑ejfv: Antibody–drug conjugate
- pembrolizumab: Immune checkpoint inhibitor
- Mechanism of action:
- enfortumab vedotin‑ejfv: Targets Nectin-4 on tumor cells, delivers cytotoxic monomethyl auristatin E (MMAE)
- pembrolizumab: Blocks programmed cell death protein 1 (PD-1), restoring T-cell anti-tumor activity
- Route of administration: Intravenous (enfortumab vedotin‑ejfv and pembrolizumab)
- Dosing Schedule:
- enfortumab vedotin‑ejfv
- 1.25 mg/kg IV on Day 1 and Day 8 Q3W × 4 cycles (preoperative, ~3 months)
- Day 1 and Day 8 Q3W × 5 cycles (postoperative, ~4 months)
- Total treatment duration: ~7 months
- pembrolizumab
- 200 mg IV on Day 1 Q3W × 4 cycles (preoperative, ~3 months)
- Day 1 Q3W × 13 cycles (postoperative, ~9 months)
- Total treatment duration: ~1 year
- enfortumab vedotin‑ejfv
- Target population: Cisplatin-eligible adults with localized MIBC
KEYNOTE‑B-15/EV-304 Phase III trial Study Design (NCT04700124)
- Study Type: Global, randomized, open-label, perioperative, active-controlled phase III trial
- Population: 808 adults with cisplatin-eligible MIBC
- Comparator: Standard perioperative cisplatin-based chemotherapy
- Treatment Regimen: enfortumab vedotin‑ejfv + pembrolizumab administered in the neoadjuvant and adjuvant settings around radical cystectomy
- Follow Up Duration:
- Primary EFS and OS assessments planned up to ~68 months
- pCR assessed up to ~47 months
- Estimated study completion: December 2026 (data reporting through ~2027)
- Key Endpoints:
- Primary: EFS
- Secondary: OS and pCR rate
Efficacy Outcomes
- Primary Endpoint Results: EFS Significantly improved versus standard chemotherapy
- Secondary Endpoint Results: OS and pCR favored, follow-up ongoing
- OS: Significant improvement, data remain immature
- pCR Rate: High rates at cystectomy
Regulatory Milestones
- Approval Status: Not yet approved; FDA decision pending
- Regulatory Pathway: Under FDA review following positive phase III KEYNOTE-B15/EV-304 results in cisplatin-eligible MIBC
- Regulatory Review: Based on statistically significant improvements in EFS, OS, and pCR rates compared with standard perioperative cisplatin-based chemotherapy
Safety
- Overall AE Burden: Manageable; consistent with known profiles of enfortumab vedotin‑ejfv and pembrolizumab
- Grade ≥3 Events: Reported at rates consistent with prior experience
- Delayed Surgery: Minimal impact
- Delayed Adjuvant Treatment: Minimal
- Treatment-related deaths: Rare
- Notable toxicity patterns:
- enfortumab vedotin‑ejfv: Peripheral neuropathy, skin reactions, hyperglycemia
- pembrolizumab: Immune-mediated events (e.g. colitis, hepatitis, endocrinopathies)
- Combined therapy: Additive but manageable
Key Clinical Implications
✔ Establishes a potentially new perioperative option for cisplatin-eligible MIBC patients
✔ Improves EFS and OS versus standard chemotherapy
✔ Achieves higher pCR rates
✔ Manageable safety profile consistent with known toxicities
✔ Potential to redefine perioperative standard of care
Bottom Line
Phase III KEYNOTE-B15 results suggest that perioperative enfortumab vedotin‑ejfv + pembrolizumab is a potentially new treatment option for cisplatin-eligible MIBC, demonstrating efficacy with an acceptable safety profile.
Sources:
- Fahey, C. C., Clark-Garvey, S., Porten, S., Kamat, A. M., Taylor, J. A., Kim, W. Y., & Milowsky, M. I. (2025). Mechanistic insights and future directions for enfortumab vedotin in urothelial carcinoma: Highlights from the 10th Annual Leo & Anne Albert Institute for Bladder Cancer Care and Research Symposium. Current Oncology, 32(5), 278. https://doi.org/10.3390/curroncol32050278
- Merck & Co., Inc. (2025, December 17). KEYTRUDA® (pembrolizumab) plus Padcev® (enfortumab vedotin-ejfv) significantly improved event-free survival, overall survival and pathologic complete response rates for cisplatin-eligible patients with muscle-invasive bladder cancer when given before and after surgery. Merck. https://www.merck.com/news/keytruda-pembrolizumab-plus-padcev-enfortumab-vedotin-ejfv-significantly-improved-event-free-survival-overall-survival-and-pathologic-complete-response-rates-for-cisplatin-eligible-pa/
- U.S. National Library of Medicine. (n.d.). A phase 3, randomized, open-label study to evaluate perioperative enfortumab vedotin plus pembrolizumab (MK-3475) versus neoadjuvant gemcitabine and cisplatin in cisplatin-eligible participants with muscle-invasive bladder cancer (KEYNOTE-B15 / EV-304) (ClinicalTrials.gov Identifier: NCT04700124). ClinicalTrials.gov. https://www.clinicaltrials.gov/study/NCT04700124