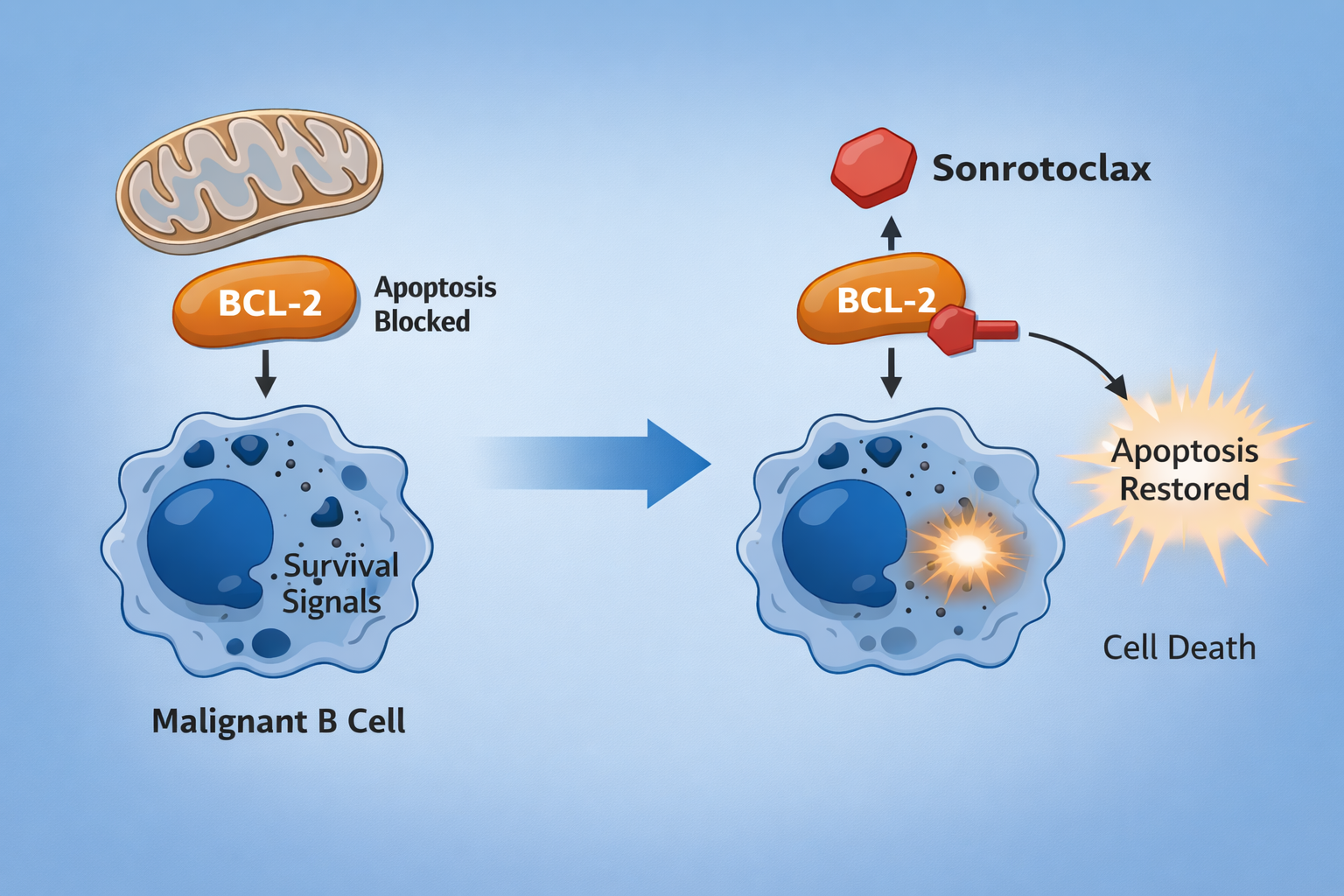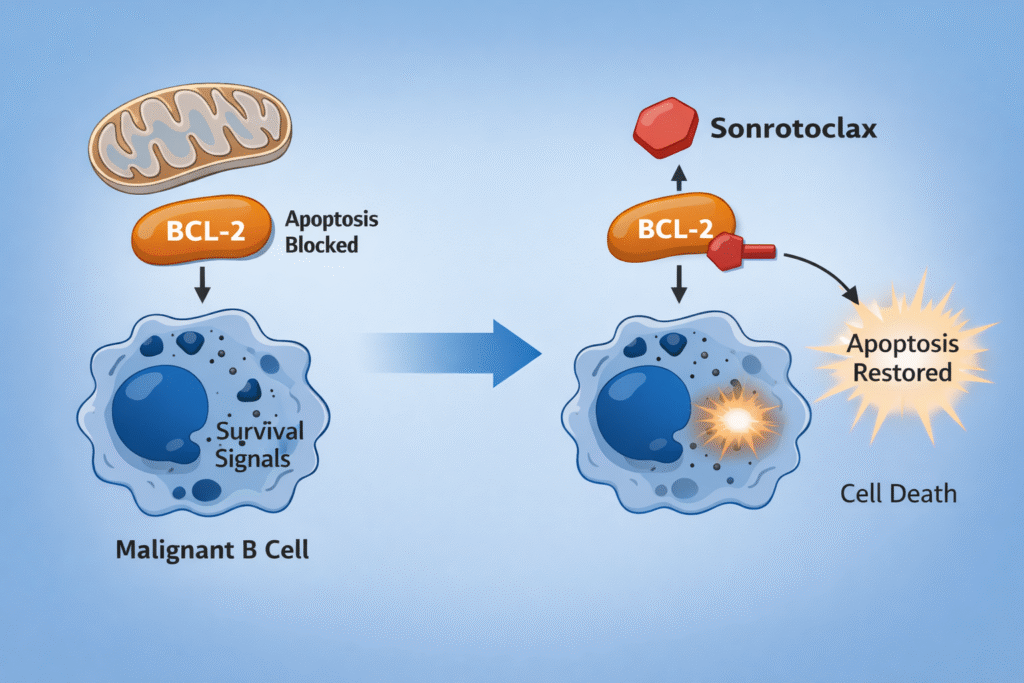
Sonrotoclax (BGB-11417) is a next generation, highly selective B-cell lymphoma 2 (BCL-2) inhibitor under investigation for patients with relapsed or refractory mantle cell lymphoma (R/R MCL), including those previously treated with Bruton tyrosine kinase (BTK) inhibitors. Early data from the phase I/II BGB-11417-201 study demonstrate clinically meaningful activity with a manageable safety profile in a heavily pretreated population, supporting U.S. FDA Priority Review consideration.
Clinical Takeaway
Preliminary phase I/II data indicate that sonrotoclax has single-agent activity in R/R MCL, including patients previously treated with BTK inhibitors. As a selective oral BCL-2 inhibitor, sonrotoclax demonstrates a manageable safety profile in early analyses. While these findings are derived from a non-randomized study with limited follow-up, they support continued clinical development and regulatory evaluation in this high-unmet-need population.
Drug Profile & Mechanism
- Agent: sonrotoclax (BGB-11417)
- Class: small-molecule BCL-2 inhibitor
- Mechanism of action: Selectively inhibits the anti-apoptotic protein BCL-2, restoring mitochondrial apoptosis in malignant B cells while minimizing off-target BCL-xL inhibition
- Route of administration: oral
- Dosing Schedule: Once daily dosing with step-up ramp-up (exact dosing levels vary by cohort, fixed dose is not available)
- Target population: Adults with R/R MCL, including patients previously treated with BTK inhibitors
BGB-11417-201 Study Design (NCT05471843)
- Study Type: Phase I/II, open-label, multicenter, single-arm
- Population:
- R/R MCL
- Heavily pretreated; majority with prior BTK inhibitor exposure
- Comparator: n/a (single-arm study)
- Treatment Regimen: monotherapy administered orally once daily following dose-escalation/ramp-up
- Follow Up Duration:
- n/a; follow-up immature at time of reporting
- Primary and Key Secondary Assessments:
- Primary: Safety and tolerability; determination of recommended phase II dose (RP2D)
- Secondary: overall response rate (ORR), duration of response (DoR), progression-free survival PFS), pharmacokinetics
- Estimated study completion: n/a, ongoing at time of data release
- Key Endpoints:
- Primary: Safety, dose limiting toxicities, RP2D
- Secondary: ORR, complete response (CR) rate, DoR, PFS
Efficacy Outcomes
- Primary Endpoint Results:
- n/a (safety focused in phase I)
- Secondary Endpoint Results: High overall response rates observed, including complete responses, in BTK-inhibitor-pretreated patients (per ASH 2025 abstract and OncLive reporting)
- Overall Survival (OS): n/a
- Hazard ratio for death: n/a
- pCR Rate: n/a in hematologic malignancy trial context
Regulatory Milestones
- Approval Status: not yet FDA approved
- Regulatory Pathway: FDA Priority Review (based on phase I/II data)
- Regulatory Review: Priority Review granted for treatment of R/R MCL
Safety
- Overall AE Burden: manageable and consistent with BCL-2 inhibitors
- Grade ≥3 Events: reported but limited; cytopenias most common
- Treatment-related deaths: none reported in available data
- Notable toxicity patterns:
- cytopenias
- low observed rates of tumor lysis syndrome (TLS) with ramp-up-dosing
- favorable selectivity profile compared with earlier BCL-2 inhibitors
Key Clinical Implications
✔ Demonstrated meaningful single-agent activity in BTK-inhibitor-refractory MCL
✔ Oral administration supports outpatient feasibility
✔ Selective BCL-2 targeting may mitigate off-target toxicity
✔ Early efficacy supports regulatory advancement despite non-randomized design
✔ Potential future backbone for combination strategies in MCL
Bottom Line
Sonrotoclax represents a promising next-generation BCL-2 inhibitor with early evidence of efficacy in a high unmet need R/R MCL population. While long term outcomes and randomized data are pending, the observed response rates, manageable safety profile, and FDA Priority Review status support sonrotoclax as a potentially meaningful addition to the evolving MCL treatment landscape.
Sources:
- Blood. (2025). Sonrotoclax (BGB‑11417) monotherapy in patients with relapsed/refractory mantle cell lymphoma (Abstract 663). Blood, 146(Supplement 1). https://doi.org/10.1182/blood‑2025‑663
- BeOne Medicines Ltd. (2025, November 26). U.S. FDA grants priority review to sonrotoclax for the treatment of relapsed or refractory mantle cell lymphoma [Press release]. BeOne Medicines. https://ir.beonemedicines.com/news/us‑fda‑grants‑priority‑review‑to‑sonrotoclax‑for‑the‑treatment‑of‑relapsed‑or‑refractory‑mantle/786f1dad‑0c9f‑492e‑9fce‑f9ceadd24989
- ClinicalTrials.gov. (2025). Study of BGB‑11417 monotherapy in participants with relapsed or refractory mantle cell lymphoma (NCT05471843). U.S. National Library of Medicine. https://clinicaltrials.gov/study/NCT05471843
- Fabbricatore, R. (2025, November 26). Sonrotoclax earns FDA priority review in R/R mantle cell lymphoma. CancerNetwork. https://www.cancernetwork.com/view/sonrotoclax‑earns‑fda‑priority‑review‑in‑r‑r‑mantle‑cell‑lymphoma
- Doherty, K. (2025, December 8). Sonrotoclax displays activity in BTK inhibitor–pretreated R/R MCL. OncLive. https://www.onclive.com/view/sonrotoclax‑displays‑activity‑in‑btk‑inhibitor‑pretreated‑r‑r‑mcl
- Wang, M. (2025, December 23). Sonrotoclax (BGB‑11417) monotherapy in patients with R/R MCL previously treated with a BTK inhibitor: Results from a Phase 1/2 study. OncLive. https://www.onclive.com/view/sonrotoclax‑bgb‑11417‑monotherapy‑in‑patients‑with‑r‑r‑mcl‑previously‑treated‑with‑a‑btk‑inhibitor‑results‑from‑a‑phase‑1‑2‑study